The role of motility and chemotaxis on tumor penetration and colonization
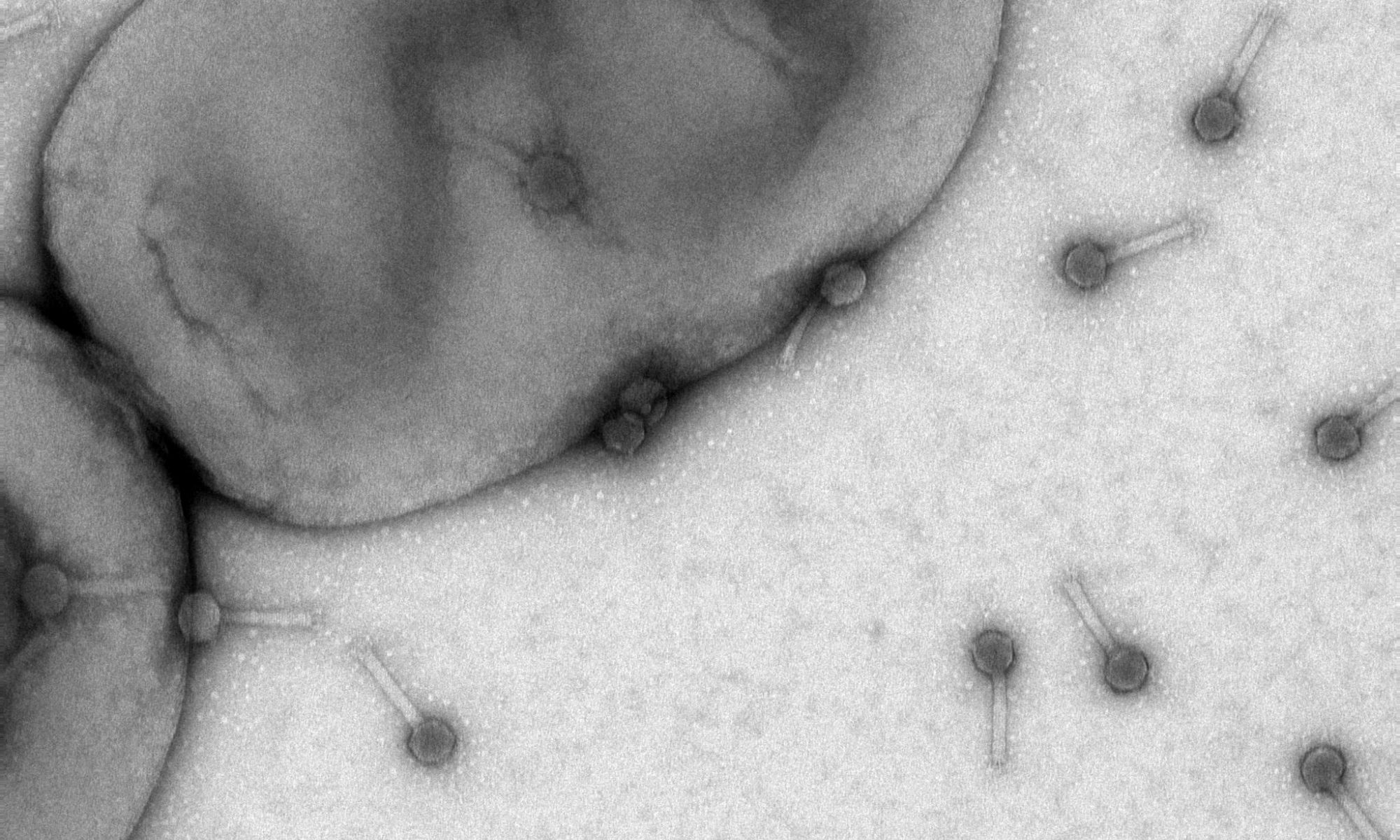
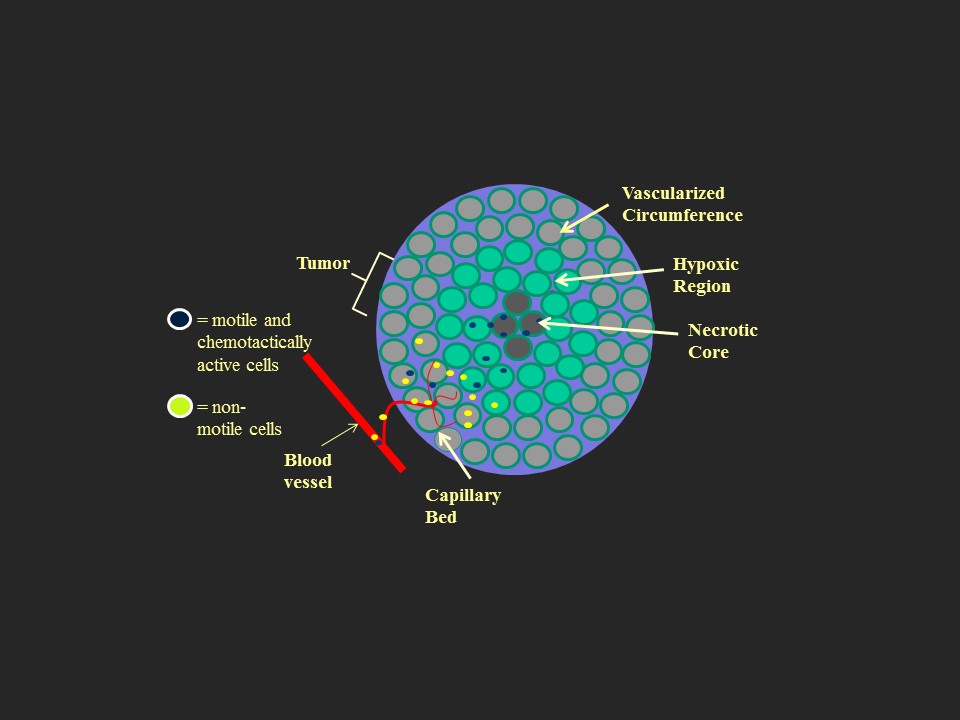
Salmonella enterica serovar Typhimurium is able to colonize tumor tissue and inhibit tumor cell growth. A mutagenized and genetically engineered derivative of S. Typhimurium 14028s is attenuated in host virulence and accumulates preferentially in tumors with a tumor-to-normal tissue ratio of 1000:1. We are using a variety of different tumor models to simultaneously analyze the expression of eukaryotic genes in response to bacterial tumor colonization and the expression of bacterial genes in response to the tumor environment. This research aims to specifically study the role of flagellar motility, chemotaxis towards tumor-generated attractants, and biofilm formation in tumors with the overarching goal to enhance bacteria-tumor interaction and, consequently, tumor penetration and eradication.
References:
K.M. Broadway & Scharf B.E. (2019) Salmonella Typhimurium as an anticancer therapy: recent advances and perspectives. Current Clinical Microbiology Reports 6(4), 225-239.
K.M. Broadway, Suh S., Behkam B, Scharf B.E. (2017) Optimizing the restored chemotactic behavior of anticancer agent Salmonella enterica serovar Typhimurium VNP20009. Journal of Biotechnology 251, 76-83. ( DOI: 10.1016/j.jbiotec.2017.04.006)
S.L. Coutermarsh-Ott, Broadway K.M., Scharf B.E., Allen I.C. of Salmonella enterica serovar Typhimurium VNP20009 and VNP20009 with restored chemotaxis on 4T1 mouse mammary carcinoma progression. (2017) Oncotarget, 8(20), 33601-33613. (DOI: 10.18632/oncotarget.16830)
K.M. Broadway, Denson E.A., Jensen R.V., Scharf B.E. (2015) Rescuing chemotaxis of the anticancer agent Salmonella enterica serovar Typhimurium VNP20009. Journal of Biotechnology 211, 117-120. (DOI:10.1016/j.jbiotec.2014.07.006)
K.M. Broadway, Modise T., Jensen R.V., Scharf B.E. (2014) Complete Genome Sequence of Salmonella enterica serovar Typhimurium VNP20009, a strain engineered for tumor targeting. Journal of Biotechnology 192, 177-178. (DOI:10.1016/j.jbiotec.2015.07.010)